Research Partners
Group Research Partners The Giacomantonio Immuno-Oncology Research Group leads the clinical-translational development of Sona’s Targeted Hyperthermia Therapy™ (THT), bridging decades of cancer immunology research with first-in-human application. Their preclinical work—including peer-reviewed studies showing that THT can convert immune-resistant “cold” tumors into immune-active “hot” tumors—laid the foundation for clinical translation. In 2025, the group led the first-in-human early feasibility study of THT in immunotherapy-resistant melanoma, generating the first clinical evidence that Sona’s CTAB-free, uniquely biocompatible gold nanorods can shrink tumors, induce apoptotic cancer cell death, reveal tumor neoantigens, and engage immune responses. Their work also identified more than 16 procedural and device refinements that are now informing next-phase clinical development.
Research Study
Evaluation of SONA’s Gold Nanoparticle-Mediated Photothermal Therapy in Combination with Intralesional Immune Modulation as a novel Immuno-Oncology strategy for Colorectal Cancers, Skin Cancers and Breast Cancers

Sona CMO:
Dr Carman Giacomantonio, MD, MSc., FRCSC
Dr. Carman Giacomantonio is a surgeon and surgical oncologist at the QEII Health Sciences Center and a professor of surgery at Dalhousie University. He is also the Chief of Surgical Services at CEHHA in Truro, NS. Dr. Giacomantonio’s research interests include cancer metastases, specifically breast, melanoma, and colorectal cancer. He has conducted several studies on the impact of surgical procedures on the tumor microenvironment and subsequent development of cancer metastases, as well as ways to mitigate this risk through targeted therapies.
Dr. Giacomantonio has published several notable papers, including “Core needle biopsy of breast cancer tumors increases distant metastases in a mouse model” and “Reduced tumorigenicity of B16-F10 mouse melanoma cells transfected with mycobacterial antigen 85A”. He has also contributed to guidelines on the use of cytoreductive surgery and HIPEC in patients with peritoneal surface malignancy.
Dr. Giacomantonio serves on several advisory boards, including the Medical Advisory Board for Colorectal Cancer Canada.
Overall, Dr. Carman Giacomantonio is a highly accomplished surgeon and researcher with a focus on cancer metastases and targeted therapies. His contributions to the field have been significant, and he continues to serve on advisory boards and conduct research to improve cancer treatment and outcomes.
Dr. Carman Giacomantonio is serving as PI for Sona’s study. The study aims to evaluate the ability of the therapy to act as a catalyst to generate immune responses and provide data that will form part of any future regulatory submissions in support of the development of its Targeted Hyperthermia Therapy. The study is innovative and marks the beginning of Sona delivering on the ‘mountain of data’ they committed to developing in support of their planned regulatory submissions for human clinical trial approvals. The results of the study are anticipated to provide valuable insights into the development of Targeted Hyperthermia Therapy, a photothermal cancer therapy that uses therapeutic heat to treat solid cancer tumors. The use of nanotechnology in cancer therapy is a rapidly growing field, and the study is an exciting development in the search for more effective and targeted cancer treatments.

Barry Kennedy, PhD
Barry Kennedy is a dedicated scientist with a passion for advancing human health and well-being. With two decades of experience spanning academic and industrial laboratories, he has devoted his career to pushing the boundaries of scientific knowledge. Currently, he holds the position of Principal Research Scientist in the Giacomantonio Immuno-Oncology Research Laboratory in the Department of Pathology at Dalhousie University. In this role, he plays a vital part in advancing research while fostering a culture of innovation within the institution. His work integrates research, translational efforts, and strategic collaborations with the biotechnology sector.
Dr. Kennedy’s science commenced with a Ph.D. in the Department of Biochemistry and Molecular Biology at Dalhousie University, where he investigated aberrant lipid trafficking and mitochondrial dysfunction during neurodegeneration. Building upon this foundation, he embarked on postdoctoral fellowships that explored deeper into the strategic manipulation of mitochondrial metabolism to enhance the efficacy of oncolytic viral immunotherapies.
As a Research Scientist and Biomarker Lead at IMV, a clinical-stage biopharmaceutical company specializing in a novel class of cancer vaccines, Dr. Kennedy provided scientific leadership, developed standard operating procedures, and engaged in strategic discussions with thought leaders. He collaborated closely with preclinical development teams to craft robust biomarker strategies and partnered with the clinical operations team to ensure scientific alignment with operational objectives during trials.
Dr. Kennedy has secured significant funding through grants and strategic partnerships with the biotechnology sector. This funding has facilitated the publication of over 30 peer-reviewed papers and garnered recognition at local, national, and international conferences. His passion for science is solid, and he is committed to leveraging his skills and expertise to make a meaningful impact on human health and well-being.
Publications

Targeted intra-tumoral hyperthermia using uniquely biocompatible gold nanorods induces strong immunogenic cell death in two immunogenically ‘cold’ tumor models – Frontiers- Sona Nanotech
Front. Immunol., 12 January 2025
Sec. Cancer Immunity and Immunotherapy
Introduction: Hyperthermia is an established adjunct in multimodal cancer treatments, with mechanisms including cell death, immune modulation, and vascular changes. Traditional hyperthermia applications are resource-intensive and often associated with patient morbidity, limiting their clinical accessibility. Gold nanorods (GNRs) offer a precise, minimally invasive alternative by leveraging near-infrared (NIR) light to deliver targeted hyperthermia therapy (THT). THT induces controlled tumor heating, promoting immunogenic cell death (ICD) and modulating the tumor microenvironment (TME) to enhance immune engagement. This study explores the synergistic potential of GNR-mediated THT with immunotherapies in immunogenically ‘cold’ tumors to achieve durable anti-tumor immunity.
ASCO - American Society of Clinical Oncology
At the 2025 American Society of Clinical Oncology (ASCO) meeting in Chicago, Sona Nanotech’s Chief Medical Officer, Dr. Carman Giacomantonio, — presented a poster featuring breakthrough preclinical findings from Sona’s gold nanorod-based Targeted Hyperthermia Therapy™ (THT). The presentation showcased how THT enables precise intratumoral heating, stimulates immune engagement, and may offer new treatment pathways for patients with resistant cancers.
Sona’s associated publication in Frontiers in Immunology — which formed the scientific foundation for the ASCO submission — has now achieved:
- 9K total views putting it in the top 13% of all viewed articles
- 1.5K downloads
- 9 citations
These metrics position the publication among the top-performing articles within the journal group, reflecting strong and growing scientific interest in Sona’s platform. ASCO also provided valuable opportunities for engagement with key opinion leaders and industry stakeholders, further validating the impact of Sona’s progress in advancing novel cancer therapies.

Additional Research Links
2023 | Abscopal responses in patients with metastatic melanoma involving skin and subcutaneous tissues treated with intralesional IL2 plus BCG
D Vidovic, LK Helyer, S Pasternak, CA Giacomantonio
Frontiers in Oncology 13, 1160269
2022 | Cutaneous Breast Cancer Metastasis Is Effectively Treated With Intralesional Interleukin-2 and Imiquimod: A Case Report and Brief Literature Review
A Drohan, D Vidovic, PJ Barnes, CA Giacomantonio, LK Helyer
Frontiers in Oncology 12, 877014
2021 | Case report: combined intra-lesional IL-2 and topical imiquimod safely and effectively clears multi-focal, high grade cutaneous squamous cell cancer in a combined liver and …
D Vidovic, GA Simms, S Pasternak, M Walsh, K Peltekian, J Stein, …
Frontiers in Immunology 12, 678028
McConnell YJ, Giacomantonio CA.
Malignant triton tumors _ complete surgical resection and adjuvant radiotherapy associated with improved survival.
J Surg Oncol. 2012 Jul 1:106(1):51-6.
Tarrant, J.P., Walsh, M.J., Blanchard, M.C., Lee, T.D.G., Hoskin, D.W., and Giacomantonio, C.A.
Reduced tumorigenicity of B16-F10 mouse melanoma cells transfected with mycobacterial antigen 85A.
Int. J. Oncol., 2004; 25: 1693-1699.
Read More About Targeted Hyperthermia Therapy
- Preclinical Small Animal Study Success
- First THT Application: Colorectal Cancer Tumors
We hypothesize that the combined utilization of GNRs and precise immune modulation will lead to enhanced immune activation and potent anti- tumor responses in colorectal cancer, breast cancer, and melanoma mouse models.
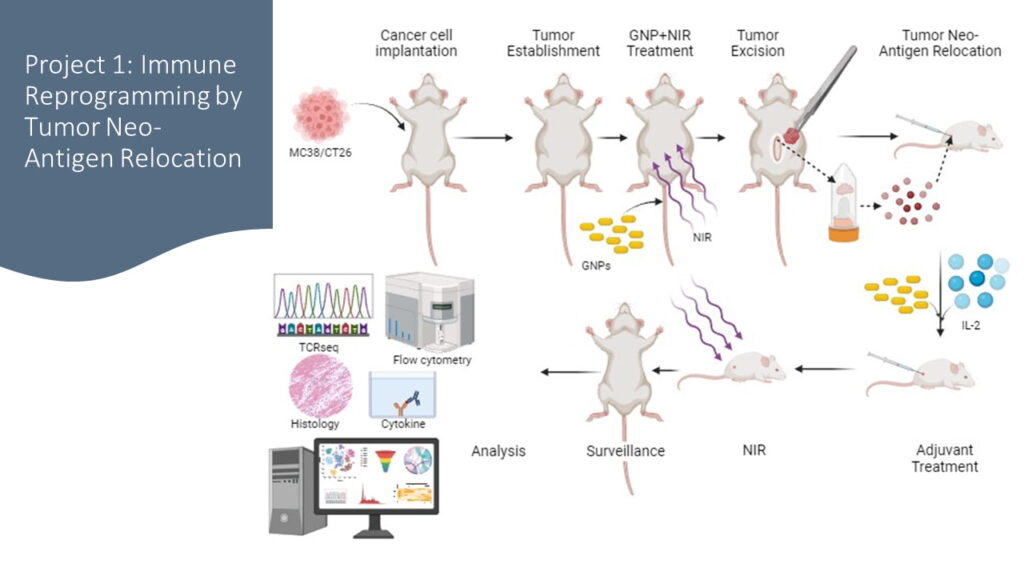
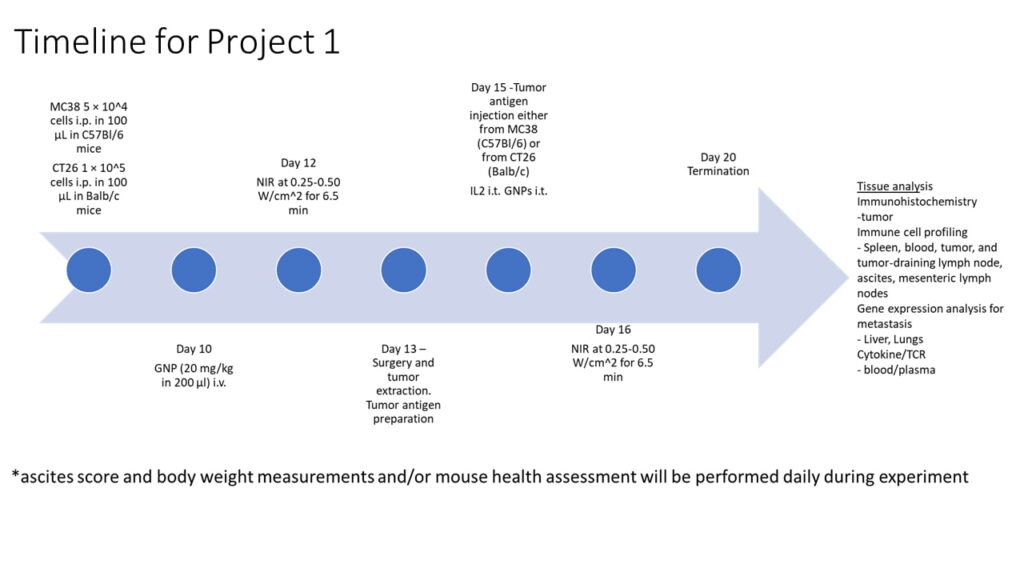
Project 1:
Immune Reprogramming through Tumor Neo-antigen
Relocation
- Harness the unique properties of GNPs to enhance immune antigen exposure
- Relocate elusive cancer antigens to highly immunogenic sites
- Unleash a personalized and potent immune response
Objective 1: Investigating i.p. Tumor Site Immune Response and Therapeutic Impact
Conducting an in-depth exploration of the immune response dynamics and therapeutic implications, specifically within the i.p. tumor site
Objective 2: Unraveling s.c. Tumor Antigen Site Immune Enhancement with IL-2
Uncover the potential of GNPs and NIR treatment, complemented by the inclusion of IL-2, in enhancing immune responses within this distinct microenvironment
Objective 3: Unveiling Molecular Pathways, Immune Checkpoint Dynamics, and Systemic Response to Tumors after Neo-Antigen Relocation Therapy
Thoroughly investigate molecular pathways and immune checkpoint dynamics at both i.p. and s.c. sites to uncover factors driving distinct therapeutic effects from combined treatments. Study systemic response post neo-antigen relocation therapy for broader insights into immune interactions beyond localized sites.
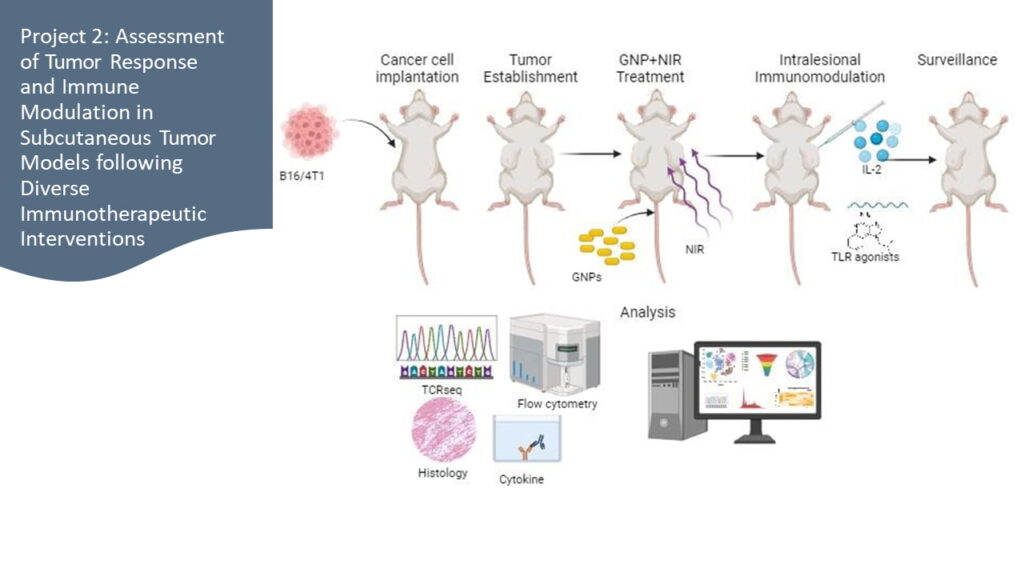
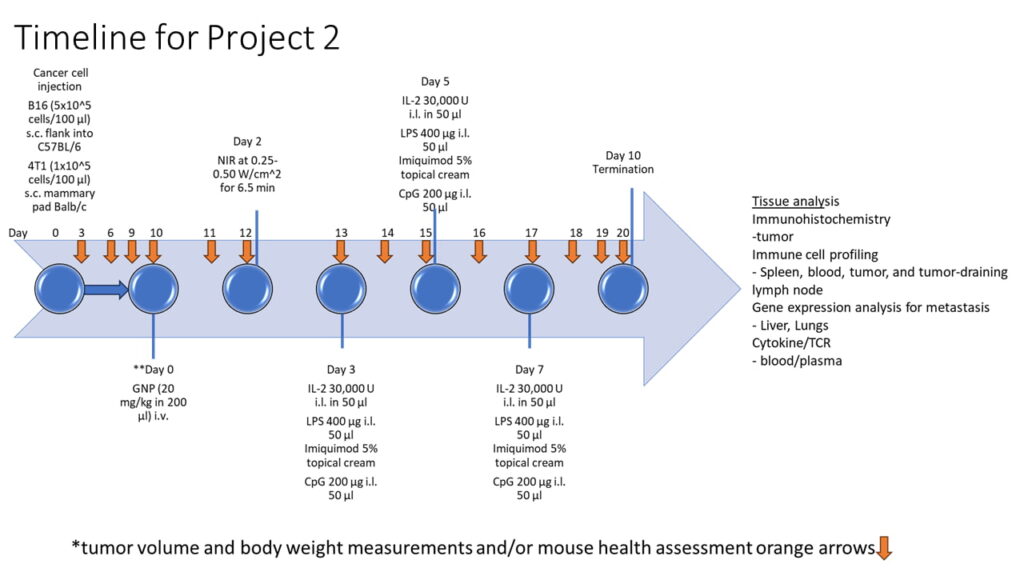
Project 2:
Evaluating Tumor Response and Immune Modulation in Subcutaneous Tumor Models after Diverse Immunotherapeutic Interventions
- Uncover the outcomes of intralesional injections of immune adjuvants, including the pivotal IL-2, combined with systemic GNPs treatment
- Develop a comprehensive immunomodulation strategy aimed at restraining tumor growth and thwarting metastasis
Objective 1: Assessing Diverse Interventions in Tumor Models
Evaluate the impact of GNPs, IL-2, and TLR agonists (TLR4, TLR7, TLR9) individually and in combinations on tumor growth, metastasis, and local/systemic immune response in 4T1 breast cancer and B16 melanoma mouse models.
Objective 2: Comprehensive Immune Analysis in Tumor Models
Analyze localized and systemic immune responses from treatments in tumor microenvironment, draining lymph nodes, spleen, and peripheral blood. Profile immune cells, cytokines, and systemic effects associated with interventions in 4T1 breast cancer and B16 melanoma mouse models.
Objective 3: Probing Molecular Pathways in Tumor Models
Investigate molecular mechanisms governing therapeutic effects, including immune cell activation, gene expression patterns, and genomic traits. Uncover intricate pathways driving observed outcomes from diverse treatments in 4T1 breast cancer and B16 melanoma mouse models.
Additional Research Links
2023 | Abscopal responses in patients with metastatic melanoma involving skin and subcutaneous tissues treated with intralesional IL2 plus BCG
D Vidovic, LK Helyer, S Pasternak, CA Giacomantonio
Frontiers in Oncology 13, 1160269
2022 | Cutaneous Breast Cancer Metastasis Is Effectively Treated With Intralesional Interleukin-2 and Imiquimod: A Case Report and Brief Literature Review
A Drohan, D Vidovic, PJ Barnes, CA Giacomantonio, LK Helyer
Frontiers in Oncology 12, 877014
2021 | Case report: combined intra-lesional IL-2 and topical imiquimod safely and effectively clears multi-focal, high grade cutaneous squamous cell cancer in a combined liver and …
D Vidovic, GA Simms, S Pasternak, M Walsh, K Peltekian, J Stein, …
Frontiers in Immunology 12, 678028
McConnell YJ, Giacomantonio CA.
Malignant triton tumors _ complete surgical resection and adjuvant radiotherapy associated with improved survival.
J Surg Oncol. 2012 Jul 1:106(1):51-6.
Tarrant, J.P., Walsh, M.J., Blanchard, M.C., Lee, T.D.G., Hoskin, D.W., and Giacomantonio, C.A.
Reduced tumorigenicity of B16-F10 mouse melanoma cells transfected with mycobacterial antigen 85A.
Int. J. Oncol., 2004; 25: 1693-1699.
Read More About Targeted Hyperthermia Therapy
- Preclinical Small Animal Study Success
- First THT Application: Colorectal Cancer Tumors