Sona's THT Leverage Its Uniquely Biocompatible Gold Nanorods
Sona Nanotech’s Targeted Hyperthermia Therapy™ (THT) is a next-generation precision cancer treatment that uses CTAB-free, uniquely biocompatible gold nanorods (GNRs) to deliver controlled, localized heat directly into solid tumors. This precise thermal activation induces apoptotic cancer cell death, reveals tumor antigens, and helps activate the immune system.
THT is designed to minimize damage to healthy tissue while overcoming the limitations of traditional cancer treatments and immunotherapy resistance. In 2025, THT demonstrated compelling early human efficacy in immunotherapy-resistant melanoma, achieving an 80% overall response rate and 60% complete responses in a first-in-human feasibility study, with a favourable safety and tolerability profile.


Uniquely biocompatible

Unlocking the potential of in-vivo medical applications

How does THT work?
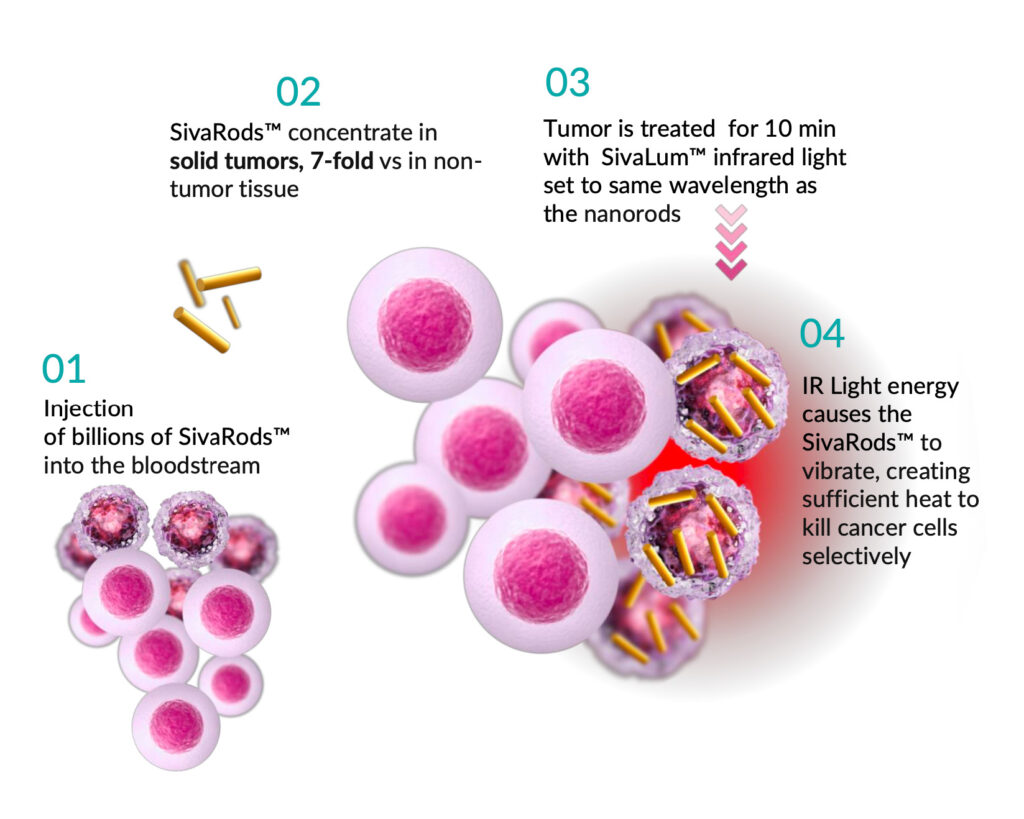
Sona’s patented, CTAB-free, uniquely biocompatible gold nanorods (GNRs) are delivered directly into solid tumors via intratumoral injection. When activated by near‑infrared (NIR) light tuned to their same wavelength, the GNRs gently heat tumors from the inside, triggering apoptotic cell death* and revealing tumor neoantigens that help the immune system better engage to fight the tumor cancer.
- Precise Delivery: Gold nanorods are injected into the tumor.
- Activation by Infrared Light: NIR light is applied via a hand-held laser which passes non-thermal light energy that penetrates up to ~2.5cm of tissue and is absorbed by the GNRs.
- Localized Heating: The gold nanorods convert the absorbed light to therapeutic hyperthermic heat, inducing apoptotic cancer cell death while minimizing damage to surrounding healthy tissue.
- Immune Engagement: Destroying cancer cells but also exposes tumor neoantigens, which can stimulate the patient’s immune system to recognize and attack cancer cells, including those previously hidden from immune detection
*Apoptotic Cell Death:
When a cell actively self-destructs in a controlled manner. In so doing, the dying cells become more “visible” to immune cells due to the altered antigen presentation which can cause the immune system to engage and attack the cancer
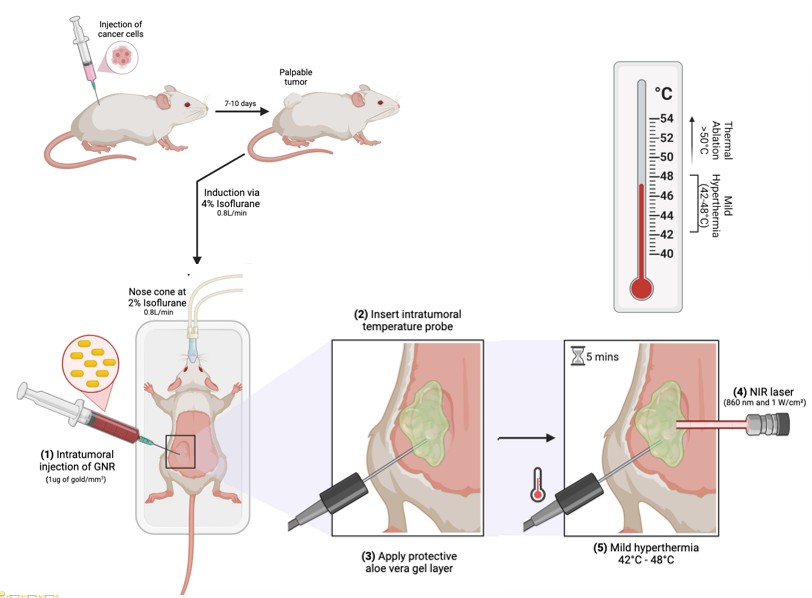
Sona’s THT Involves Two Injections Of GNRs Followed By ~5 Minutes Of Sona’s Near-infrared Laser Energy Which GNRs Convert To Heat
Clinical Progress & Recent Milestones
Early Feasibility Study Results (2025)
Sona Nanotech completed its first-in-human early feasibility study evaluating Targeted Hyperthermia Therapy™ (THT) in patients with immunotherapy-resistant melanoma. The study demonstrated compelling early human efficacy, including:
– 80% overall response rate
– 60% complete responses in biopsied tumors
– Favourable safety and tolerability, with no THT-related serious adverse events
– 16+ procedural and device refinements supporting ongoing optimization
These results represent the first clinical evidence that CTAB-free gold nanorod-based hyperthermia can shrink tumors and engage immune responses in humans.
Study Design
Participants received two THT treatments one week apart, with the study evaluating:
– Safety and tolerability
– Tumor response
– Local immune engagement
– Device and procedural performance
Preclinical Success
Prior to human testing, THT demonstrated strong preclinical efficacy across multiple cancer models including melanoma, and colorectal and triple negative breast cancer. Studies showed that combining THT with immunotherapy can convert immune-resistant “cold” tumors into “hot” tumors, enhancing immune activation and improving treatment outcomes.
Preclinical Foundation
Prior to human testing, THT demonstrated strong preclinical efficacy across multiple cancer models including melanoma, and colorectal and triple negative breast cancer. Studies showed that combining THT with immunotherapy can convert immune-resistant “cold” tumors into “hot” tumors, enhancing immune activation and improving treatment outcomes.
Next Steps in Clinical Development
Following the favourable early feasibility clinical outcomes:
- Sona has filed an Investigational Testing Authorization (ITA) with Health Canada for a clinical trial for which it has already received research ethics board approval.
- Histology analysis and immune-infiltrate assessment remain ongoing.
- A pilot study of ~30–40 patients is planned for 2026, pending regulatory clearance.
- The pilot study will refine treatment parameters, evaluate multi-centre deployment, and generate data to inform pivotal trial design.
Next Gen Laser
Sona’s Next Generation Laser is a custom-engineered, near-infrared light device designed specifically for use with Sona’s gold nanorod (GNR) technology in Targeted Hyperthermia Therapy (THT). This device has undergone extensive validation and verification to ensure it reliably delivers consistent energy output with robust safety features, meeting the stringent requirements of clinical trials and regulatory bodies.
- Optimized wavelength (860 nm): Penetrates up to one inch into tissue, allowing precise targeting of tumors from the surface or within body cavities
- Consistent, reliable energy delivery: Rigorously validated for clinical use, with built-in safety features to ensure dependable performance
- Non-invasive approach: Enables access to tumors with harmless near infrared light without the need for surgical procedures, supporting minimally invasive cancer therapy.

Unlike surgical lasers that cut or ablate tissue, Sona’s laser gently transmits non-thermal near-infrared light, which is absorbed by the injected gold nanorods. These nanorods then convert the light into heat, selectively destroying cancer cells from the inside out while sparing healthy tissue. This approach not only treats tumors directly but also has the potential to stimulate the immune system, supporting Sona’s vision for innovative, minimally invasive cancer therapies

Pioneered by
Len Pagliaro, PhD
- 24 years of experience
- Successful commercialization of biotechnology products, services, and licensing
- Completed prestigious assessment program at the US Nanotechnology Characterization Laboratory (Established by the FDA and the National Cancer Institute)
Sona GNRs in targeted hyperthermia may create the opportunity to treat cancer without doing significant harm to healthy cells
Current Cancer Treatments are risky, expensive and can do harm
- Chemotherapy and radiotherapy are non-selective in their destruction of cells;
- Advanced therapies are expensive;
- Surgery is risky.
Advantages & Future Potential
Minimally Invasive
THT delivers precise, localized thermal activation within tumors, reducing collateral damage and minimizing side effects compared with systemic chemotherapy, radiation, or surgical intervention.
Immune System Activation:
By inducing selective apoptotic cancer cell death and revealing tumor neoantigens, THT helps engage the immune system — a mechanism supported by both preclinical studies and early human findings in immunotherapy-resistant melanoma.
Versatile Platform
Sona’s gold nanorod (GNR) platform is being advanced for multiple solid tumor types. Insights from the early feasibility study will inform expansion into additional indications as part of future clinical development.
Safety & Biocompatibility
Sona’s CTAB-free, uniquely biocompatible gold nanorods eliminate the toxicity concerns associated with legacy gold nanorod technologies. Independent validation from the US NIH’s National Cancer Institute’s Nanotechnology Characterization Laboratory (NCL) reassessed Sona’s GNRs as stable and free of detectable endotoxins.
Clinical Integration
As a locally delivered, immune-activating therapy, THT is designed to complement existing cancer treatments. Early clinical results suggest the potential for meaningful integration alongside immunotherapies in patients with treatment-resistant disease.
How does THT work?

- Targets heat directly to the tumor;
- Uses IV injection of GNRs to heat the tumor from the inside;
- Achieves hyperthermia instead of ablation destroying cancerous cells selectively;
- Sona GNRs are inert and do not use toxic CTAB.
Targeted Hyperthermia Therapy™ (THT)
Medical device with two components:
- Gold nanorods for injection
- Infrared light source
Heating tumors does the following:
- Stimulates immune system
- Kills cancer cells
- Increases tumor perfusion
- Shrinks tumors
Healthy cells withstand heat stress, up to 52°C, typical with ablation therapies.
Not damaged by hyperthermia’s 44°C
THT destroys cancer cells while healthy cells can stay undamaged
SivaRods™ heat to 44°C
Selectively kills cancer cells
Works from the inside of the tumor out
Heat shock protein (HSP) synthesis used

Cancer cells are more sensitive to heat
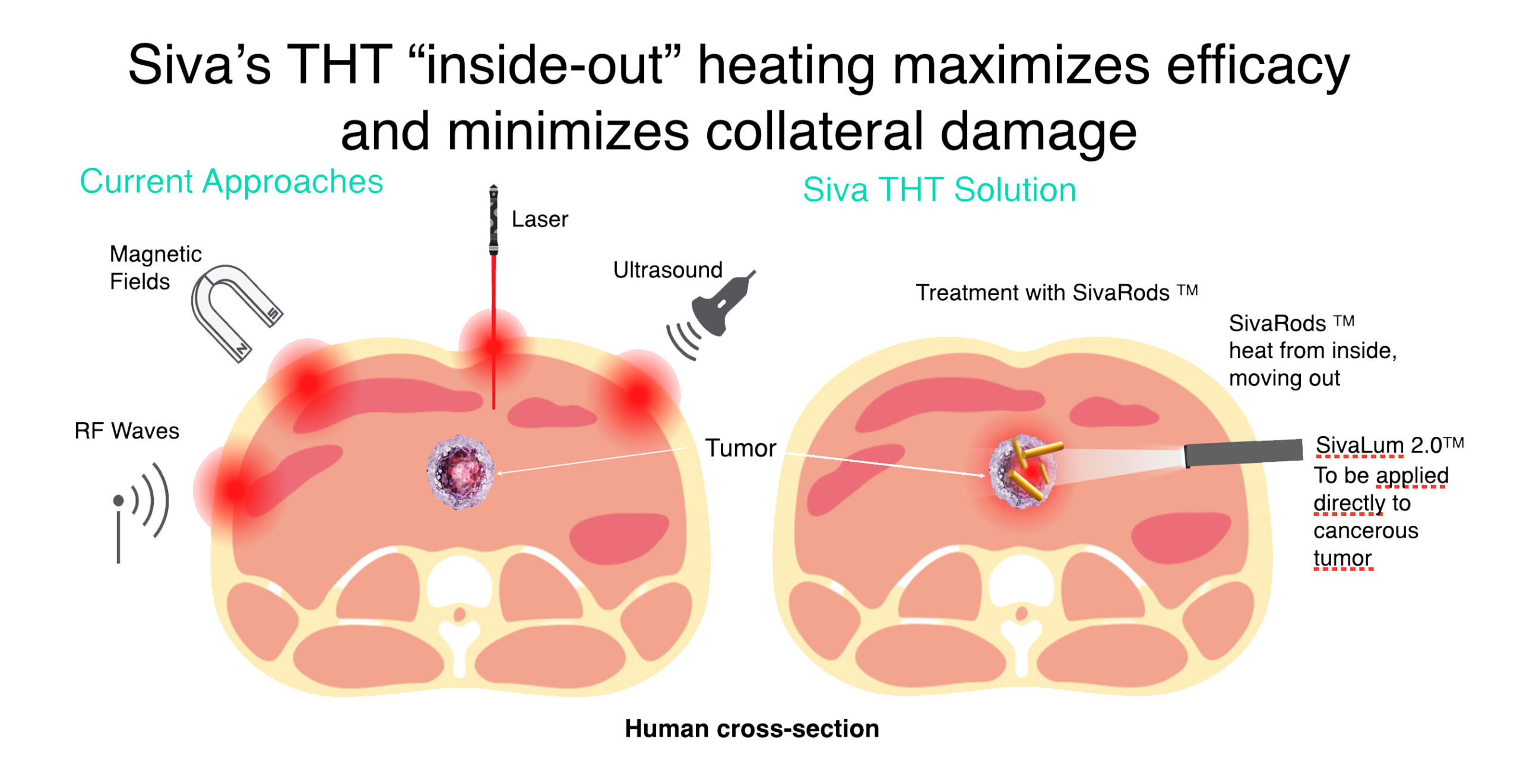
Siva’s Unique Differentiator:
Selective ‘Hyperthermia’ Minimizes Collateral Damage
- THT photothermal cancer therapy using GNRs will address current treatment issues.
- Siva's THT "inside-out" heating maximizes efficacy and minimizes collateral damage.

Hyperthermia
Blue Rare (115°F) i.e. 46°C
Hyperthermia doesn’t harm normal cells

Ablation
Medium (134°F) i.e. 57°C

Ablation therapies ‘cook’ proximate cells
- Siva has had major success in reducing tumors in small animal studies.
- Eliminated tumors in mice in 4 weeks
- Validated by Nanotechnology Characterization Laboratory study and report.
THT photothermal cancer therapy using GNRs will address current treatment issues
Key Issue
Using gold ‘in vivo’ is understood to be safe
Long-term effects of GNRs treated with toxic CTAB are unknown.
Sona GNR Advantage
Sona’s proprietary CTAB-free GNRs have shown no toxicity
Confirmed by third party and in-house testing

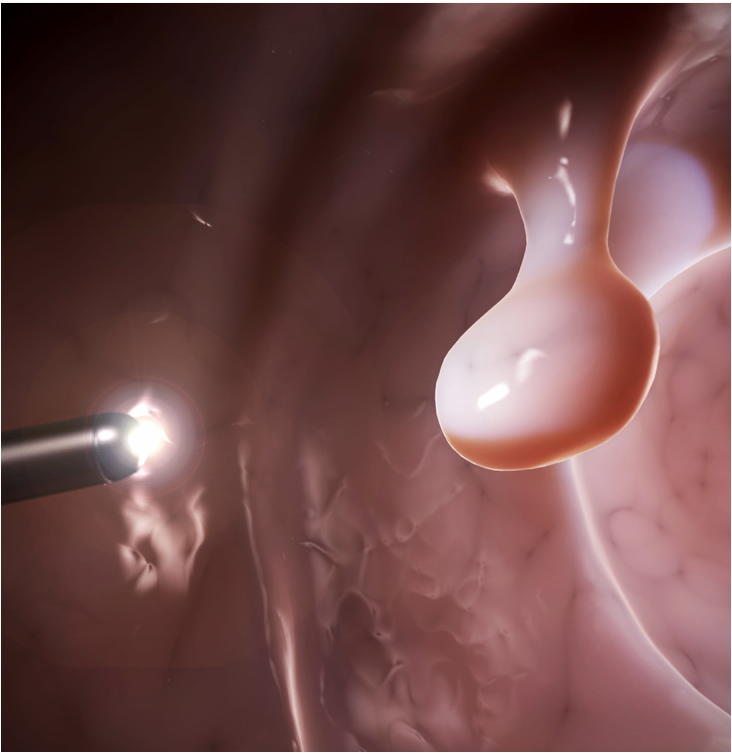



First THT Application: Colorectal Cancer Tumors
Why is THT uniquely suited for colorectal cancer treatment?
- Alternatives diminish quality of life
- Significant market
- Outpatient procedure within current workflow
- Effective for solid tumors
- Early detection is possible
- THT can be integrated with “watch and wait” approach
- Low metastatic index
Treatment Benefits:
- Minimally invasive
- Targeted treatment
- Enhances success of other cancer therapies
- Easy to use
- Affordable
Read More About Targeted Hyperthermia
- First Patient Dosed in Early Feasibility Study Trial of Its Cancer Therapy
Road to Commercialization
Potential future clinical studies to provide multiple valuation catalysts.
Additionnal Publications